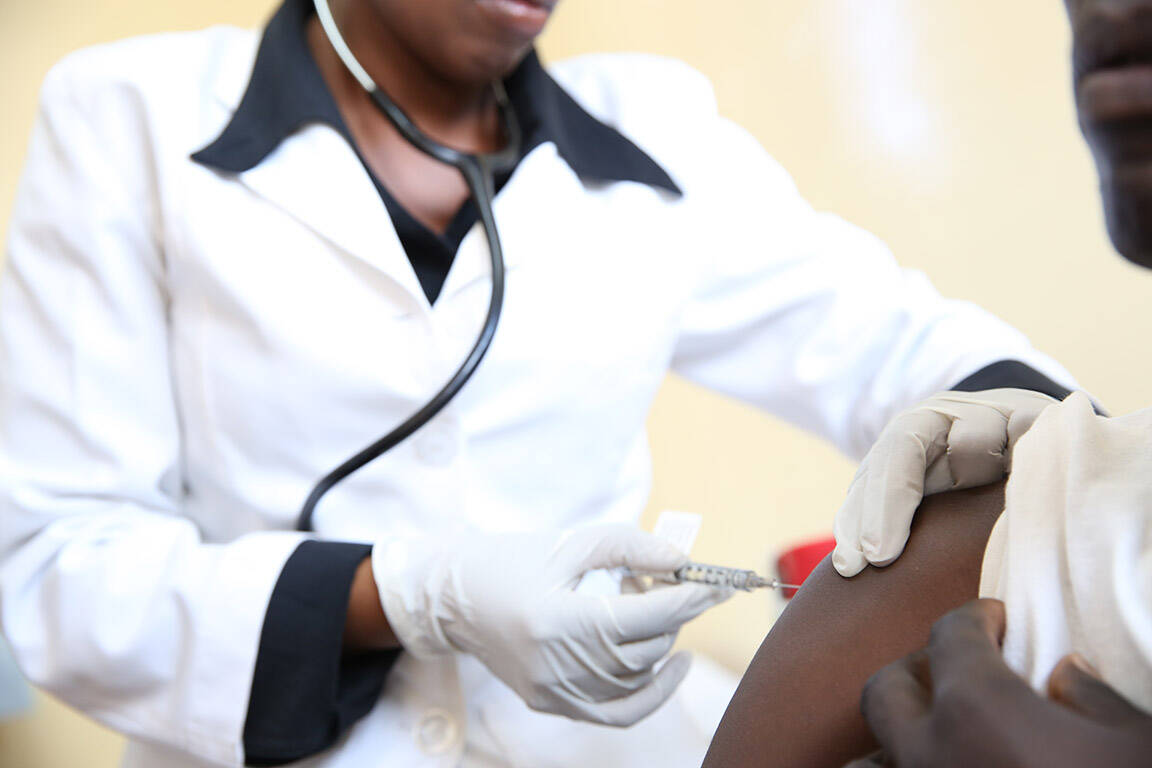
In 2021, the ETEC Vaccine Efficacy study completed a phase I safety study in Zambia on progressively younger age groups (6–23 months). With no safety issues identified, a phase IIb study has begun in The Gambia, which will assess the ability of ETVAX® to prevent ETEC-associated diarrhoea in infants aged 6–18 months.
The related ETEC ETVAX project is developing a new formulation of ETVAX® that will be better suited to young children, as well as a convenient device for administering the vaccine. This new all-in-one formulation will be evaluated in an EDCTP-funded phase III trial in Zambia, involving infants 6–18 months of age. Ultimately, the project aims to recruit 7500 infants, and the results will pave the way to an application for WHO prequalification of ETVAX®, facilitating its introduction into countries in sub-Saharan Africa.
Together, the projects will advance development of a vaccine projected to protect against 80–85% of the ETEC strains that affect young children in Africa.
scroll down
ETVAX® is the most advanced oral vaccine against ETEC. It contains a combination of engineered strains of E. coli that produce high levels of four proteins known to stimulate powerful immune responses, as well as a hybrid protein that combines E. coli and cholera toxins.
Enterotoxigenic E. coli (ETEC) infections cause a severe and life-threatening diarrhoea responsible for at least 50,000 deaths a year, mainly of young children. Death rates are highest in sub-Saharan Africa.
Two projects focused on prevention of harmful E. coli infections in children made excellent progress in 2021.

Advancing a key E. coli vaccine
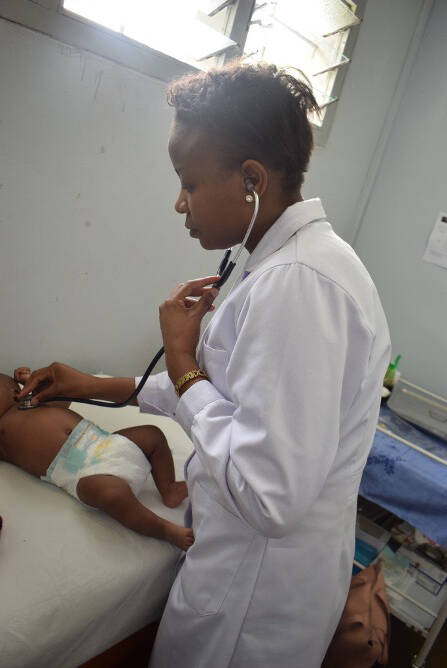
Chest examination by a physician
Project: ETEC Vaccine Efficacy study
Project lead: Mr Björn Sjöstrand, Scandinavian Biopharma Holding AB, Sweden
Countries involved: Finland, The Gambia, Sweden, United Kingdom, Zambia
Year funded: 2019
EDCTP funding: €7.4 M
Grant agreement: RIA2017S-2024
Mr Björn Sjöstrand
(Sweden)

What comes next for the projects?
Björn Sjöstrand: The enrollment of a total of 4936 children in the phase IIb trial in The Gambia will be completed before the end of June 2022 and the last child will receive the third booster dose in October 2022. A passive surveillance period for clinically significant (moderate to severe) diarrhoea episodes will follow after administration of the third dose. The new tailor-made formulation for infants and young children in LMICs will be ready before the phase III study.
The related ETEC ETVAX project is developing a new formulation of ETVAX® for children. Why is it relevant to pursue this formulation and how is this being achieved?
Björn Sjöstrand: A new and more user-friendly (partially dry) formulation of ETVAX® has been developed. To ensure that the new formulation produces an equivalent immune response to the previously tested formulation, comparative immunogenicity (immunobridging) study was initiated in 2021. Further development of a tailor-made formulation for infants and young children in LMICs is ongoing.
The advantage of the new, self-contained formulation is that it will be easy to use. The required water is already included, which facilitates the administration of the vaccine. The new formulation also has the advantage that the person administering the vaccine can choose to use a spoon, syringe, or cup for administration.
What progress was made in 2021 towards the project’s objectives?
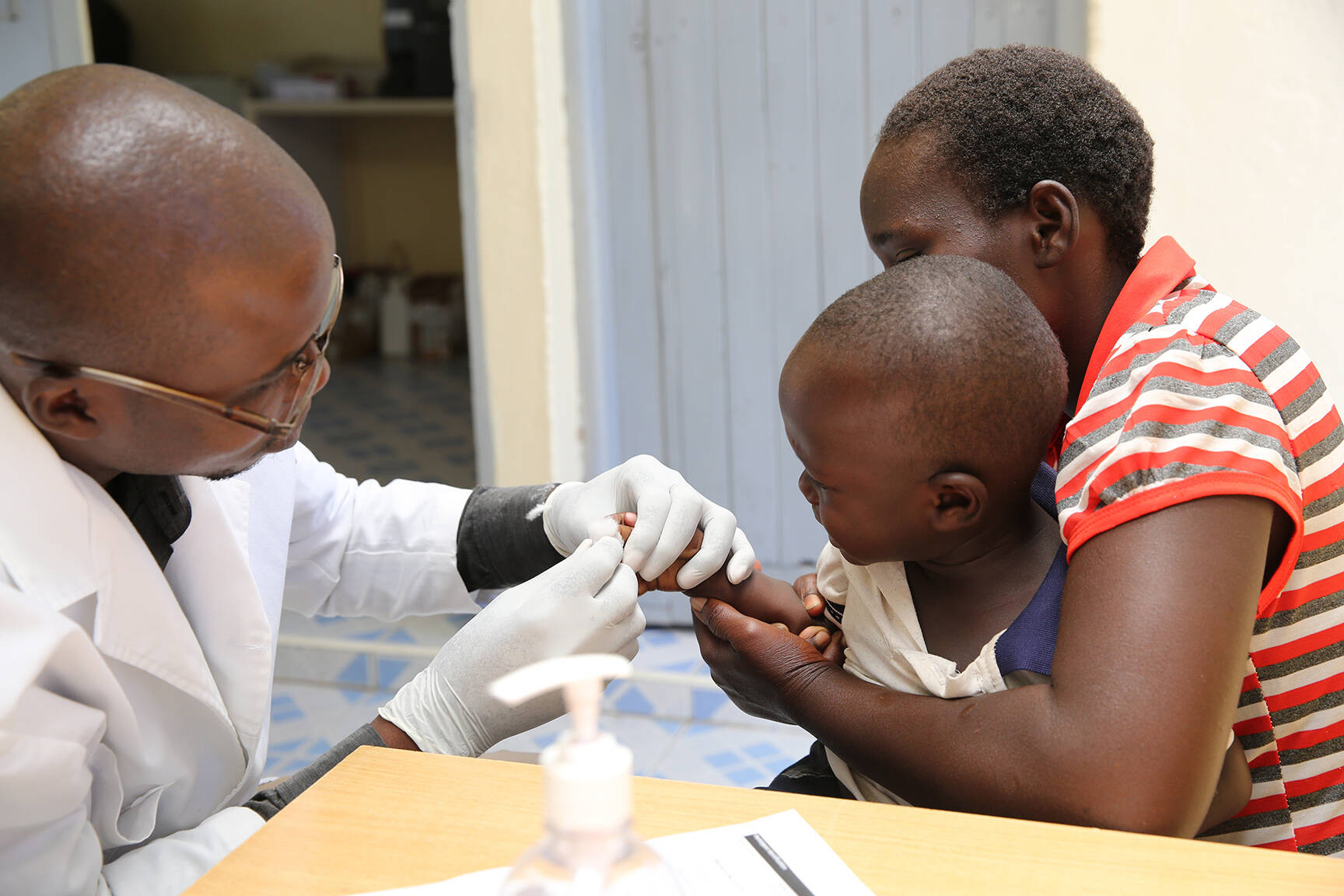
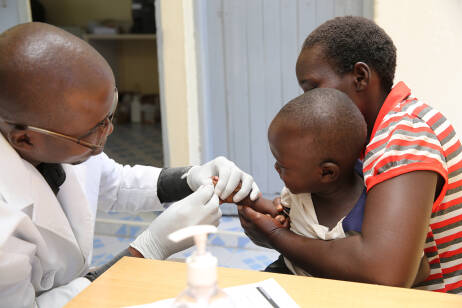
Björn Sjöstrand: The final Clinical Study Report for the phase I trial in Zambia was delivered in June and the results support the safety, tolerability and immunogenicity of ETVAX® in adults and children. It also showed a clear benefit of a third booster dose in infants and young children. In the phase IIb trial, the field sites have been equipped, lab-related technology transfer was initiated, staff have been trained, and we are proud to report that 3036 children were randomised before the end of 2021.
The ETEC Vaccine Efficacy project is collecting data on the efficacy of ETVAX® in African children. Why is this important and how will you use the data?
Björn Sjöstrand: Enterotoxigenic E. coli (ETEC) remains a major cause of diarrhoea-associated mortality and morbidity in low- and middle-income countries (LMICs). ETVAX® has strong potential to be the first vaccine with an ETEC indication. The objective of the project was to establish the safety and immunogenicity of ETVAX® and to explore the potential benefits of a third booster dose in infants and young children in Zambia, as well as to measure the protective efficacy in a phase IIb field trial in The Gambia. The two trials are designed to prepare ETVAX® for a phase III trial in Zambia.
scroll down
Project Q&A
Mr Sjöstrand with members of his team.