
index
Collaborative clinical studies and clinical trials

2014-2021
33 grants
€194.83 M

EDCTP portfolio: Tuberculosis
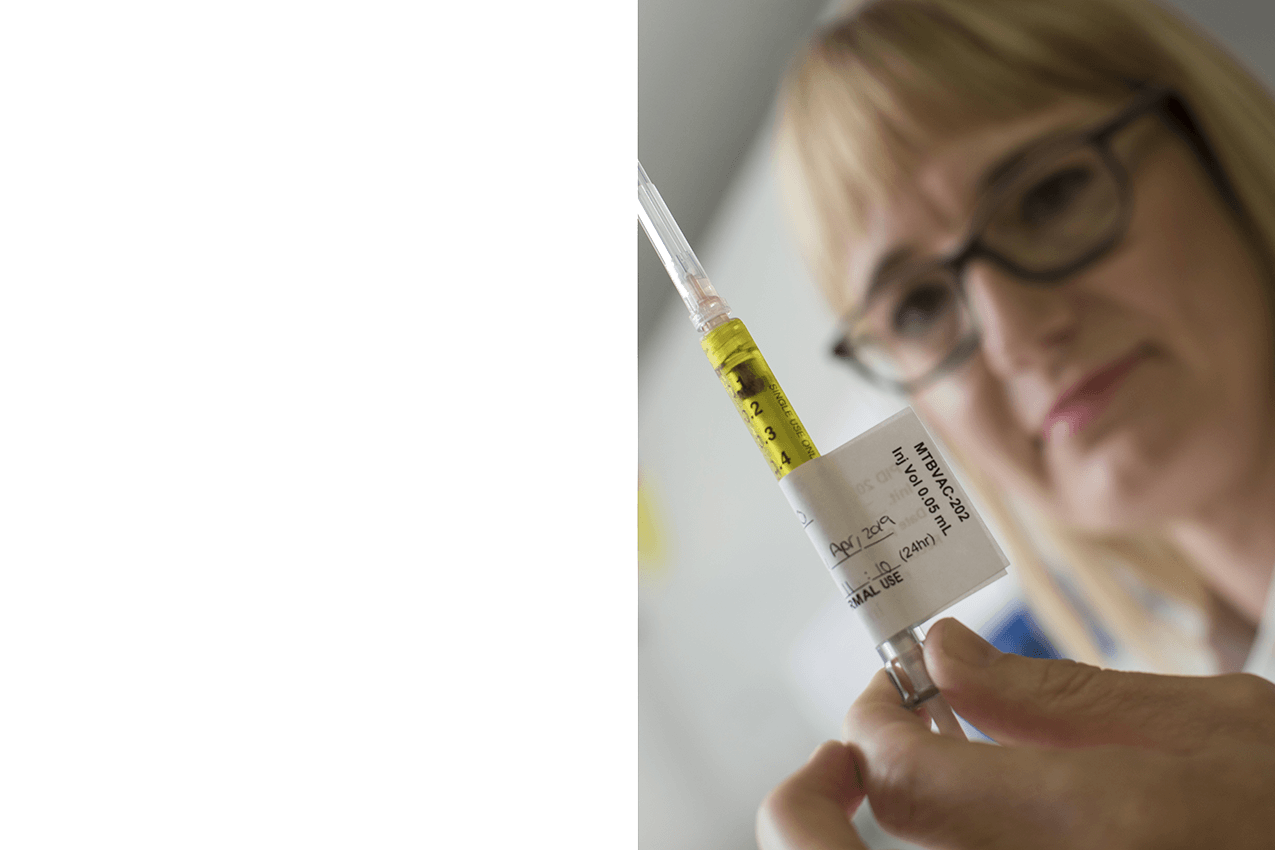

priMe: Building a successor to BCG

MTBVAC - Newborns: Better TB protection for newborns

POR TB: A new approach for TB vaccination

INTENSE-TBM: Improving treatments for TB-associated meningitis

StatinTB: Statin use to boost anti-TB therapies

CLICK TB: Advancing new options for treatment of TB

Simplici-TB: Shorter treatment for drug-resistant TB

TB-CAPT: Facilitating implementation of TB testing

TREATS: Tackling TB and HIV together

VirTUAL: Treating TB and HIV in neglected groups

PanACEA: Advancing a suite of TB drug treatments
Note: A further €26.06M for 24 grants was awarded to projects on nonintervention-specific.
33 grants
€194.83 M

2014-2021
EDCTP portfolio: Tuberculosis



